TCCV1.3 290124
Participant Information sheet
This Macmillan Cancer Support evaluation is being conducted by Edinburgh Napier University.
Purpose of Study: The purpose of this evaluation is to understand the referral process and satisfaction with your local authority transforming cancer care service (TCCS) often referred to as Improving Cancer Journey (ICJ).
Who Can Participate: If you are a health and social care professional who refers your cancer patients into the local TCC / ICJ service in Scotland you are eligible to participate?
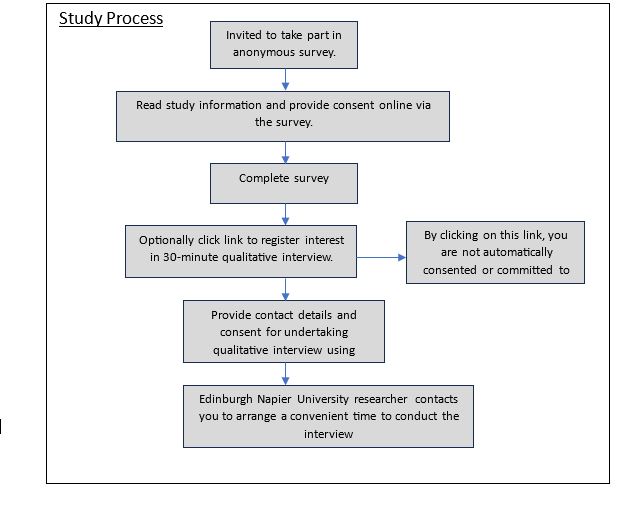
Procedure for the survey: This is an anonymous online survey followed by an optional qualitative interview with Edinburgh Napier University researchers. To participate you need to complete the consent form on the next page of the survey. No information you share electronically can be traced to you or the computer you used.. Depending upon the depth of your responses, participation time varies from 10 and 20 minutes. At the end of this survey, you will have the option to provide additional consent to undertake the qualitative interview.
Procedure for the in-depth Interviews: The qualitative interview will be undertaken using Microsoft teams. If you click the link at the end of the survey you will be taken to a Microsoft form and asked to provide consent for the qualitative interview and provide contact details to arrange a convenient time to undertake the interview. This is to maintain anonymity from the survey. The interview will be recorded with your consent.
Anonymity: The online survey will be anonymous. The in-depth interviews will be recorded and transcribed removing all potential identifiable data for analysis. All files will sit on a secure, confidential password protected computer server. All findings will be reported as a group with no ability to identify individual participation.
Risks of Being in the Study: the evaluation team do not perceive that there will be a risk to being in the study as this is about your current clinical use of the TCCS 0r ICJ service.
Potential benefits: Research participants will be contributing to the understanding of how the TCCS or ICJ service is satisfying you and your cancer patients. This will also help understand the variation or consistence across the Scotland as Macmillan Cancer Support role out the service.
As a participant please, refrain from mentioning individual names and place of work when answering the questions. This will ensure your anonymity in both the survey and the in-depth interview.
Voluntary Nature of the Study: Your participation is voluntary, and you may refuse to participate or withdraw at any time without penalty or loss of benefits to which you are otherwise entitled. You may also refuse to answer certain questions. A decision to participate or to withdraw, will not affect your professional status.
Contacts and Questions: If you have a concern about any aspect of this evaluation study, you should speak to the researcher who will do their best to answer your questions, please contact on the Research Assistant Kali Thomson K.Thompson@napier.ac.uk .
If you wish to discuss this study with an independent person, please contact: Dr. Coral Hanson, Convener, School of Health and Social Care Integrity Committee, email: c.hanson@napier.ac.uk
